

Arthritis remains the most common cause of disability in adults in the United States,1 and it is among the leading conditions causing work limitations.2 By 2030, the number of adults affected with doctor-diagnosed arthritis is projected to reach 67 million, or 25% of the adult population. Corresponding arthritis-attributable activity limitation is projected to reach 25 million, meaning that 9.3 % of all adults will be affected.3 Estimating the prevalence and burden of the various conditions that comprise arthritis and other rheumatic conditions is important for understanding the current and growing impact of these conditions on the health care and public health systems. It is equally important to identify the gaps in our understanding of these measures and target potential interventions.
Arthritis and other rheumatic conditions (AORC) comprise more than 100 diseases. What many of them have in common is that they cause pain and aching, and stiffness or swelling in or around a joint.
Defining AORC to assess their burden in the population requires considering both what is important to measure and what data sources are available, such as population surveys and administrative data. Complicating any definition is the 100+ conditions that comprise what is generally thought of as arthritis. Furthermore, population measures need to be relatively simple and perhaps different from definitions used in clinical practice, where there is the luxury of having a medical history, physical examination, and laboratory and radiographic data. The Centers for Diseases Control and Prevention (CDC) Arthritis Program has worked with other organizations to develop case definitions, based on the best available expertise, that allow many measures of population burden to be addressed in a consistent way.1
For self-reported population surveys, doctor-diagnosed arthritis is defined as a “yes” answer to the question: “Have you EVER been told by a doctor or other health professional that you have some form of arthritis, rheumatoid arthritis, gout, lupus, or fibromyalgia?” This measure aims to capture most of the major categories of arthritis and is considered valid for surveillance purposes of estimating population prevalence.2 For data sources using International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM) codes, arthritis and other rheumatic conditions (AORC) has been defined by the National Arthritis Data Workgroup using those codes, and further divided into 10 more specific subcategories defined in Arthritis and Joint Pain Codes [1]. Both measures were designed to exclude or minimize other major categories of musculoskeletal disease such as osteoporosis and generic chronic back pain although some adults with arthritis do have chronic back pain. Doctor-diagnosed arthritis likely is better for estimating what is happening in the general population, while AORC is likely better for estimating what is happening in the health care system.
While AORC occurs in children, it is difficult to get population data on them, so most of the estimates presented in this report are for adults unless otherwise noted.
Arthritis: The prevalence of arthritis in the general population is better estimated by doctor-diagnosed arthritis than by AORC [3]. Based on self-reported data, for the years 2010 to 2012, doctor-diagnosed arthritis affected an average of 52.5 million adults based on self-reported data,1 and in line with earlier projections.2 Estimates show the typical distribution of higher prevalence among females and older adults, and lower prevalence among Hispanics and Asians. Absolute estimates show that most of these adults (62%, or 32.7 million) are age 18 to 64. (Reference Table 4.1 PDF [4] CSV [5])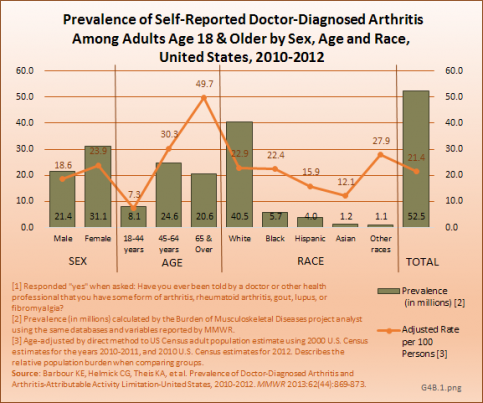
Specific types of AORC: Clinical data are needed to provide some measure of validity for estimating the prevalence of specific types of arthritis because many people are not sure what specific type of arthritis they have. Data from the National Arthritis Data Workgroup provided the following national prevalence estimates for 2005.3,4
Osteoarthritis: Osteoarthritis (OA) is the most common type of arthritis, characterized by progressive damage to cartilage and other joint tissues. Joint injury is a risk factor for most types of OA, but the majority of cases occur without a specific history of injury. Obesity is a risk factor for knee OA, and to a lesser extent for hip and hand OA. Clinical OA was estimated to affect 26.9 million in 2005(4) and is likely to have increased since then. The joints most affected with radiographic OA and symptomatic OA were hands, knees, and hips.4
Rheumatoid arthritis: Rheumatoid arthritis (RA) is the prototypical inflammatory arthritis. It is a chronic autoimmune disease that causes pain, aching, stiffness, and swelling in multiple joints, especially the hands, in a symmetrical fashion. In 2005, RA was estimated to affect 1.3 million adults(2), but that estimate has since been updated on the basis on newer data to 1.5 million.5
Gout and other crystal arthropathies: Gout is a recurrent inflammatory arthritis that occurs when excess uric acid collects in the body. Gout has been recognized for centuries and often affects the big toe. In 2005, an estimated 6.1 million adults reported having gout at some time, with 3.0 million affected in the past year.4
Systemic lupus erythematosus: Systemic lupus erythematosus (SLE) is an autoimmune disease in which the body’s immune system can attack many body systems, especially the skin, kidneys, and joints. In 2005, definite and suspected SLE was conservatively estimated to affect 322,000 people in the United States.3
Systemic sclerosis: Systemic sclerosis (SSc), or scleroderma, is an autoimmune disease that primarily affects the skin, but can affect any organ system. In 2005, SSc affected an estimated 49,000 adults.3
Primary Sjögren’s syndrome: Primary Sjögren’s syndrome (SS) is a syndrome of dry eyes, dry mouth, and arthritis. Secondary Sjögren’s syndrome can occur in association with other rheumatologic diseases such as rheumatoid arthritis and lupus. Prevalence data are very limited. In 2005, an estimated 0.4 to 3.1 million adults had SS.3
Fibromyalgia: Fibromyalgia (FM) is a syndrome of widespread pain and tenderness. The diagnosis is difficult to make, so relevant prevalence data are hard to come by. In 2005, FM was estimated to affect about 5 million adults.4
Polymyalgia rheumatica and giant cell (temporal) arteritis: Polymyalgia rheumatic (PMR) is a syndrome of sudden aching and stiffness in older adults that responds to treatment with anti-inflammatory medications (eg, corticosteroids). Giant cell arteritis (GCA), which often occurs with PMR, is a type of vasculitis that affects medium-size arteries and results in headache, vision loss, and other symptoms. In 2005, PMR was estimated to affect 711,000 people and GCA was estimated to affect 228,000 people.4
Spondylarthropathies: Spondylarthropathies (or spondylarthritides) are a family of diseases that includes ankylosing spondylitis, reactive arthritis, psoriatic arthritis, enteropathic arthritis (associated with ulcerative colitis or Crohn’s disease), juvenile spondylarthritis, and undifferentiated spondylarthritis. In 2005, spondylarthropathies affected an estimated 639,000 to 2.4 million adults ages 25 and older.3
Arthritis in children: Arthritis and other rheumatic conditions are relatively uncommon in children, although they can be particularly severe when they do occur. One estimate using significant pediatric arthritis and other rheumatologic conditions (SPARC) codes put the average annual prevalence at 294,000 for the years between 2001 to 2004, with codes for rheumatoid arthritis and other inflammatory polyarthropathies, allergic purpura, arthropathy associated with infections, other and unspecified arthropathies, polyarteritis nodosa and allied conditions, and rarer inflammatory conditions affecting 103,000.6
In recent years there has been an increase in the impact of arthritis and other rheumatic conditions on health care utilization. The rising prevalence of AORC and the resulting increase in ambulatory care visits as well as the increasing number of joint replacements and resultant increase in hospitalizations both result in increased health care utilization. As mentioned above, the AORC case definition is more appropriate to use within the health care system, so the following estimates are based on that.
AORC hospitalizations. Data from the Healthcare Cost and Utility Project (HCUP) 2011 Nationwide Inpatient Sample (NIS) estimates 6.7 million hospitalizations that are associated with a diagnosis of AORC, or 17.3% of all hospitalizations that year. Because hospitalization for osteoarthritis (OA) and rheumatoid arthritis (RA) is rare, only about 1% of hospitalizations associated with an AORC diagnoses had this as the presenting, or first, diagnosis. However, arthritis is often a contributing cause to hospitalization, particularly when associated with joint replacement surgery; therefore, most of the 6.7 million hospitalizations were associated with OA, gout, RA, and other less specific disorders. (Reference Table 4.2 PDF [6] CSV [7])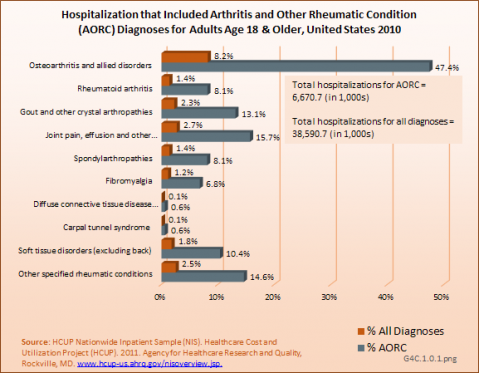
Most AORC-associated hospitalizations in 2011 occurred among women (60%), with gout the only arthritis condition where more men were hospitalized. Although people age 65 years and older comprise only 17% of the adult population, they accounted for 41% of AORC-associated hospitalizations. (Reference Table 4.3 PDF [8] CSV [9]).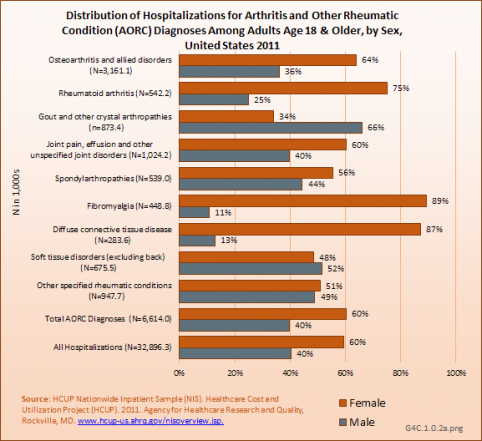
The rate of hospitalization rate per 100 people for AORC-associated hospitalizations was similar men and women. Among the 10 AORC subgroups, hospitalizations rates increased by age most significantly for osteoarthritis and other specified rheumatic conditions. AORC did not differ from all hospitalizations in hospitalization rate by sex, but did differ in the rate by age. (Reference Table 4.3 PDF [8] CSV [9])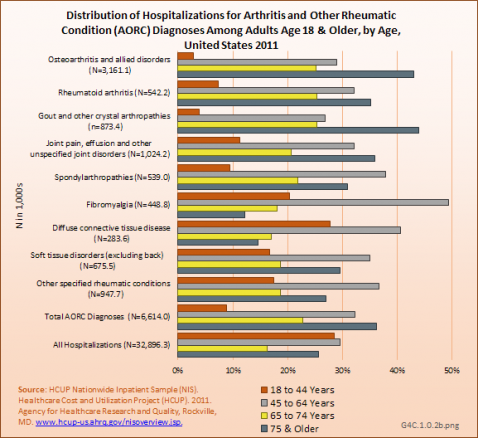
The mean length of stay for AORC-associated hospitalizations was 5 days, and it did not differ much by sex or age. Compared with all hospitalizations, AORC-associated hospitalizations averaged 1.6 days longer mean length of stay, but was even longer for those aged 18 to 44 years. Among the 10 AORC subgroups, mean length of stay was longer than the AORC average for those with gout, diffuse connective tissue disease, soft tissue disorders, and other specified rheumatic conditions. (Reference Table 4.4 PDF [10] CSV [11])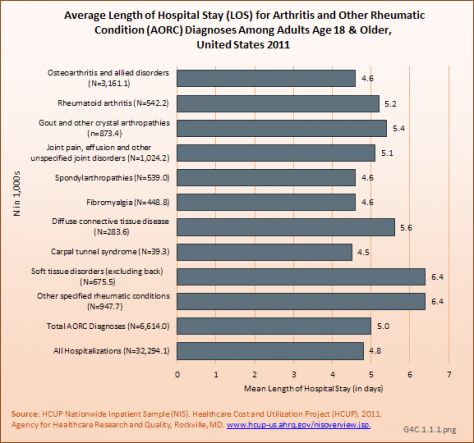
Mean hospital charges for AORC-associated hospitalizations were $5,500 higher than those for all hospitalizations, with the differences driven primarily by females and those younger than 65 years of age. Total charges for AORC-associated hospitalizations were $291 billion, comprising 23% of all hospital charges in 2011. This percentage was relatively consistent for all sex and age groups except those 18 to 44 years of age. Among the 10 AORC subgroups, osteoarthritis accounted for $135 billion, or 46% of total charges for AORC-associated hospitalizations. (Reference Table 4.4 PDF [10] CSV [11])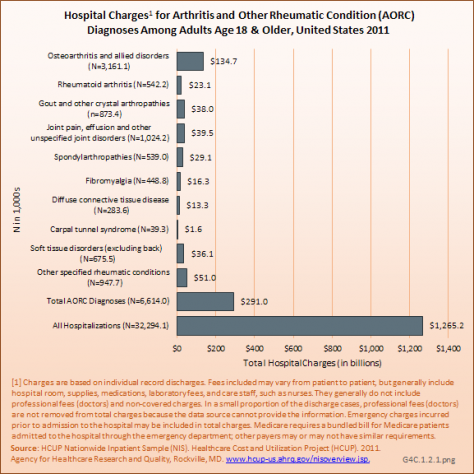
Discharge from the hospital to long-term care or home health care occurs more often among AORC-associated hospitalizations than is found among hospitalizations for all causes. This is true regardless of sex or age. Among the 10 AORC subgroups, discharge to home was more frequent and similar to that among all hospitalizations among those with fibromyalgia, diffuse connective tissue disease, and spondylarthropathies. (Reference Table 4.5 PDF [12] CSV [13]) 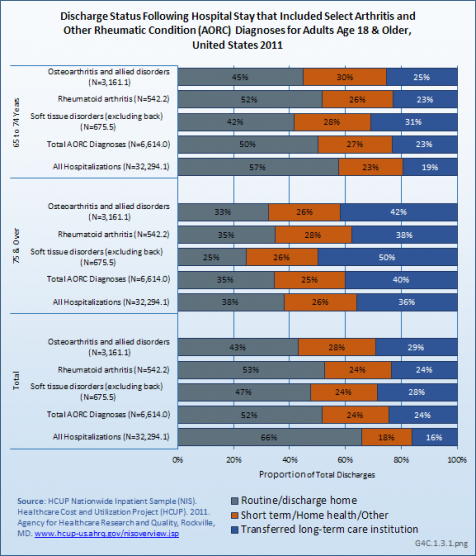
Data from the 2010 NHCS surveys on ambulatory care indicate that more than 100 million ambulatory care visits are associated with a diagnosis of AORC, or nearly 10% of all visits that year. An AORC is listed as the presenting (first) diagnosis for between 2.6% and 5.7% of all visits listed, depending on the health care site visited. Physicians’ offices accounted for 84% of all ambulatory visits, greatly exceeding emergency department or outpatient sites. Among the 10 AORC subgroups, most of the 100 million visits were associated with unspecified joint disorders and other specified rheumatic conditions; osteoarthritis was the most common specific condition. (Reference Table 4.2 PDF [6] CSV [7])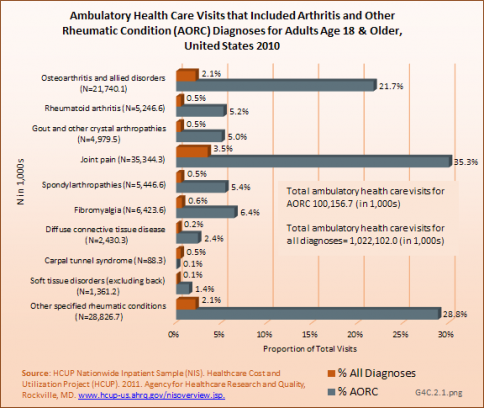
Disease burden can be measured in many ways. This is particularly important for AORC, which has a modest effect on conveniently measured outcomes like mortality, but a much larger impact on less conveniently measured outcomes important to the ability to function for most people. Such outcomes include effects on work, sports activities, health-related quality of life, independence, and ability to keep doing valued life activities. Three of these burdens, along with lifestyle factors that impact on arthritis, are addressed in the data.
Bed days are defined as spending one-half or more days in bed because of injury or illness, excluding hospitalization. Among adults with doctor-diagnosed arthritis in 2012, 22.4 million, or 9.5% of the entire adult population experienced bed days. These individuals reported an average of 24.0 bed days in the past 12 months. This is far higher than the 14.5 bed days reported by adults with any medical condition. This resulted in 537.6 million bed days overall, or 53% of the 1 trillion bed days among adults reporting any medical condition. Females and those 75 years and older had higher than average bed days. (Reference Table 4.7 PDF [14] CSV [15])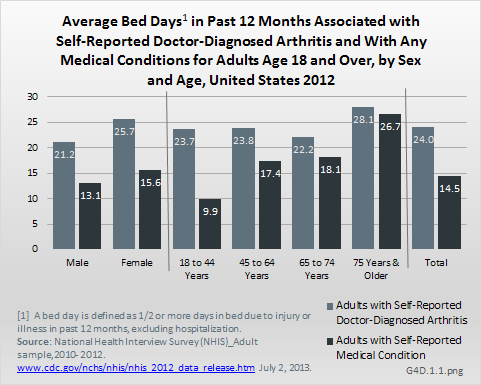
Lost work days for persons in the workforce are defined as absence from work because of illness or injury in the past 12 months, excluding maternity or family leave. Among adults with doctor-diagnosed arthritis in 2012, 12.0 million experienced lost work days. These individuals reported an average of 14.3 workdays lost in the past 12 months, far higher than the 9.9 work days reported by adults with any medical condition. This resulted in 172.1 million total work days lost due to AORC, or 33% of the 526.4 million work days lost among adults reporting any medical condition. There was little difference reported by sex in work days lost, but age was a factor, with more wor kdays lost by those aged 65 years and older. (Reference Table 4.7 PDF [14] CSV [15])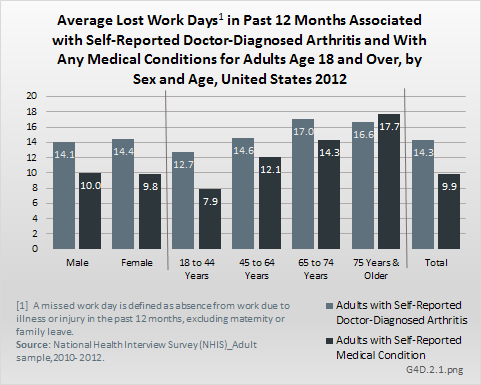
Arthritis-attributable activity limitations (AAAL) are defined by the self-reported question: “Are you now limited in any way in any of your usual activities because of arthritis or joint symptoms?” in the National Health Interview Survey [16]. Estimated doctor-diagnosed arthritis prevalence was also used to estimate that AAAL affected an average of 22.7 million adults between 2010 and 2012,1 higher than the 22 million projected to be affected by 2020 in a 2006 project.2 Estimates showed the typical distribution of higher rates among females and older adults, and lower rates among Hispanics and Asians. Absolute estimates show that most of the adults with arthritis-attributable activity limitation (13.8 million, or 61%) were under age 65 years. However, these ages comprise 83% of the US population. Thus, the rate of AAAL is higher in persons age 65 years and older. (Reference Table 4.1 PDF [4] CSV [5])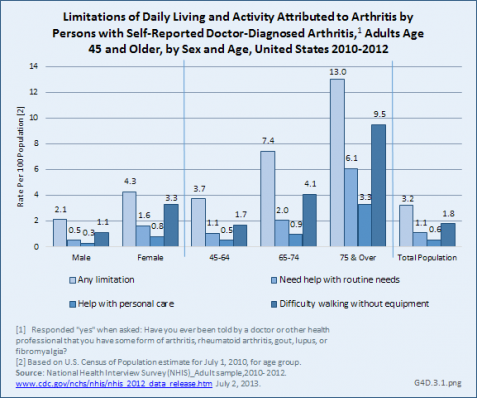
Arthritis-attributable work limitations (AAWL) are defined in reference to work for pay as: “Do arthritis or joint symptoms now affect whether you work, the type of work you do, or the amount of work you do?” In 2002, AAWL affected an estimated 30% adults (6.9 million) aged 18 to 64 years with doctor-diagnosed arthritis. Higher rates were found among those aged 45 to 64 years, women, non-Hispanic blacks, and those with low education or low income.3 Between 2010 and 2012, 3.8 million people age 18 years and older with doctor-diagnosed arthritis reported they are “unable to work now due to a health,” while 2.1 million reported they are “limited in the kind or amount of work they can do.” Arthritis conditions represented about one in five people with a work limitation from any medical condition. (Reference Table 4.6.1 PDF [17] CSV [18]; Table 4.6.2 PDF [19] CSV [20])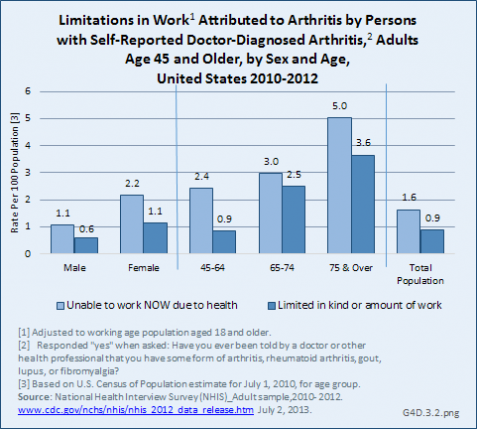
Limitations in daily living and activity (ACL) National Adult Survey data from 2010 to 2012 examined those with “any limitation” and with three specific ACL limitations—routine needs, personal care, and walking. Participants were asked to attribute their limitations to up to three medical conditions.
Among all adults with limitations, those with doctor-diagnosed arthritis naming arthritis as the cause comprised 19% of the estimated 40.8 million with “any limitation.” One-third of people with difficulty walking (32%) attributed their limitation to arthritis, while about one in four attributed limitations with personal care and routine needs to arthritis. This demonstrates the large impact of arthritis on adults with daily chronic limitations. This effect was much stronger among females than males and among older adults. Little difference was found between persons by race. (Reference Table 4.6.1 PDF [17] CSV [18]; Table 4.6.2 PDF [19] CSV [20]; and Table 4.6.3 PDF [21] CSV [22])
Among people with doctor-diagnosed arthritis (DDA) compared with those without doctor-diagnosed arthritis, Health-Related Quality of Life (HRQoL) is worse on several scales. When assessed by self-reported health status, 27% of those with DDA reported fair/poor health compared to 12% of those without DDA. The DDA group also reported a higher mean number of days in the past month with poor physical health (6.6 vs. 2.5 days), poor mental health (5.4 vs. 2.8 days), or days with limitations in usual activities (4.3 vs. 1.4 days).1
Switching to the perspective of the general population and using a different survey, the prevalence of doctor-diagnosed arthritis and of arthritis-attributable activity limitations is much higher among those with the lifestyle factors of obesity, insufficient or no physical activity, and fair/poor self-rated health. In this same survey, those with doctor-diagnosed arthritis had very high proportions of all three lifestyle factors. (Reference Table 4.8 PDF [23] CSV [24])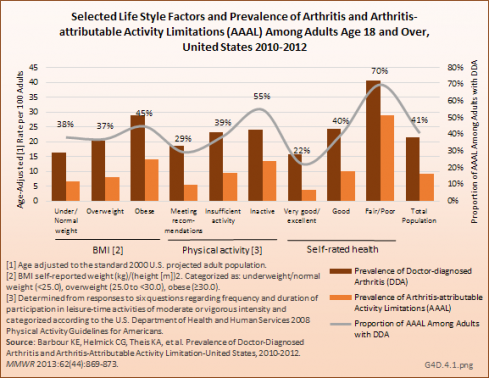
While the need for a joint replacement in a sense represents a failure of measures to prevent the occurrence or progression of joint problems, for those with the severe pain or poor function of end-stage joint problems, it can represent a life-altering “cure.” Joint replacement procedures for hips and knees are most common, but replacements have been expanding to other joint sites over the past few years. Joint replacements represent one of the fastest growing procedures in the United States.
Data are provided for both of the two national hospital discharge databases for comparison purposes. Although they vary slightly in the number of cases, overall they provide relatively consistent estimates of inpatient joint replacement procedures.
In 2010 and 2011, there were an estimated 1.3 to 1.4 million inpatient joint replacement procedures. Joint replacement procedures comprised about 3.5% of all inpatient procedures for those years. More joint replacements are performed on women than men, and 93% of the procedures are performed on knees or hips. (Reference Table 4.9 PDF [25] CSV [26])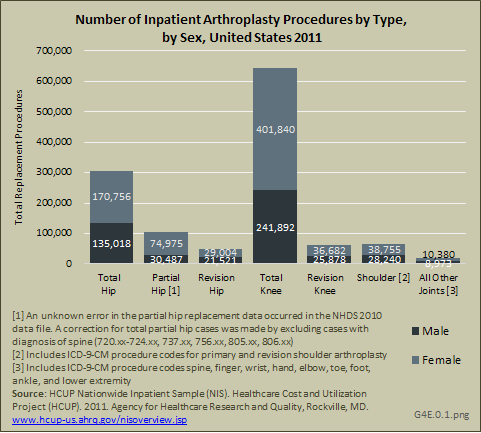
In 2010 and 2011, an estimated 706,000 to 757,000 knee replacement procedure were performed on patients in the United States, comprising 56% of all joint replacement procedures. Three in five knee replacements occurred in females. Total knee replacements far exceeded revision knee replacements, which occur when the original replacement fails or becomes infected. (Reference Table 4.9 PDF [25] CSV [26])
Estimated trends in knee replacement procedures from 1992 to 2010 or 2011 show steady increases in both total and revision knee replacements. Over the 18 years, knee replacement procedures approximately tripled, with the ratio of revisions to total remaining constant at 8% to 10%. (Reference Table 4.10 PDF [27] CSV [28]).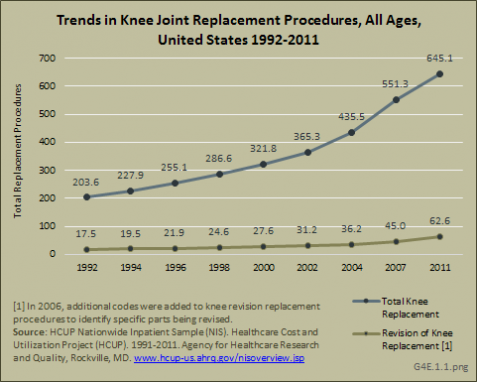
The principal diagnosis associated with total knee replacement is osteoarthritis, accounting for 95% or more of all replacements. (Reference Table 4.11 PDF [29] CSV [30]).
The mean age for both total knee and revision knee replacements was 68 years over an 18-year period. The mean age for both procedures shows a slow decline over this time period. (Reference Table 4.12 PDF [31] CSV [32])
The mean length of stay for both total and revision knee replacements has shown a remarkable decline of about 50% from a mean of nearly 9 days in 1992 to a mean of 3.5 days in 2010. (Reference Table 4.13 PDF [33] CSV [34]).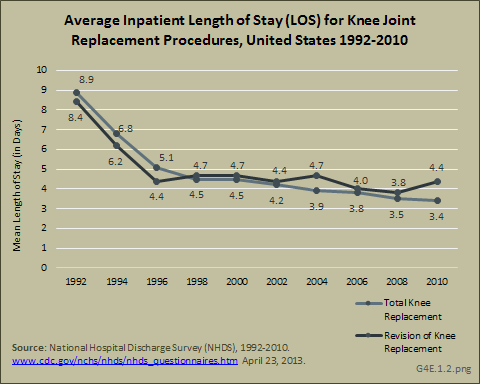
In spite of shorter hospital stays, the mean hospital charges from 1998 through 2011 showed steady increases for both knee replacements, with revision knee replacement being more expensive than total knee replacement. Total hospitalization charges for both knee replacements have more than quadrupled (in constant 2011 dollars) from $8.1 billion in 1998 to $38.5 billion in 2011. (Reference Table 4.14 PDF [35] CSV [36])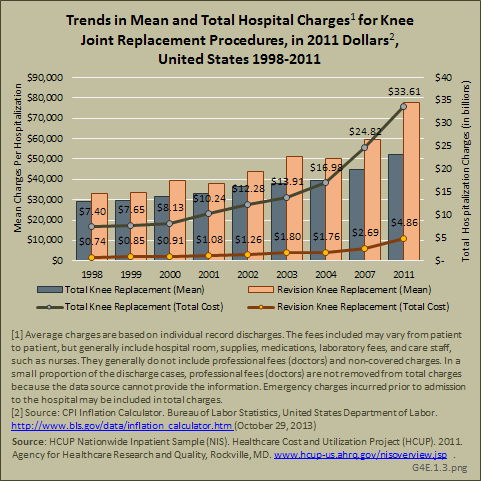
Most adults with knee replacements are routinely discharged home, with 20% to 30% discharged to long-term care. Among persons age 65 years and older, a slightly higher proportion are discharged to long-term care. (Reference Table 4.15 PDF [37] CSV [38]).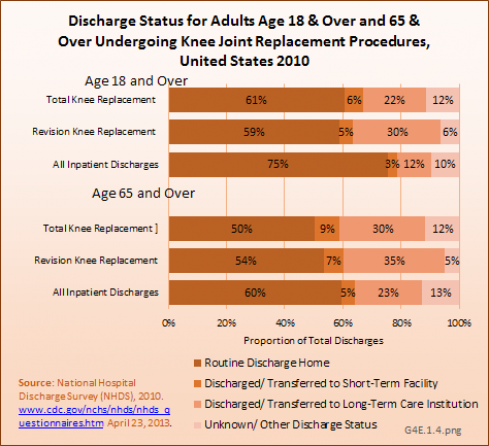
In 2010 and 2011, there were an estimated 465,000 to 512,000 hip replacement procedures, comprising 37% of all joint replacement procedures. A majority, about 63%, occurred in females. Total hip replacements occur nearly three times as frequently as partial hip replacements, and both are far more common that revision hip replacement. (Reference Table 4.9 PDF [25] CSV [26])
Estimated trends in hip replacement procedures from 1992 to 2010 or 2011 show a steady increase in all types of replacements, with total hip replacements more than doubling by 2010/2011. Growth is slowest in partial hip replacements. The ratio of revision hip to total hip replacements hovers around 20% for most years. The years 2006/2007 showed anomaly data for revision hip replacements in both data sets. (Reference Table 4.10 PDF [27] CSV [28])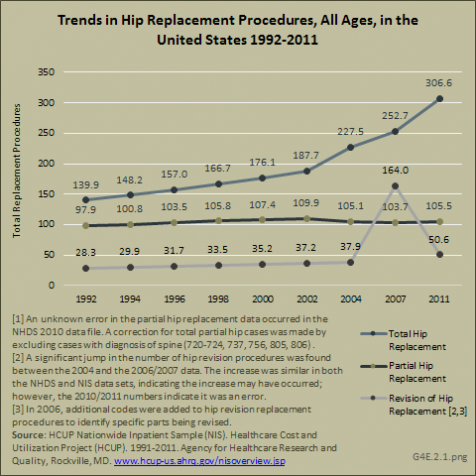
The principal diagnoses associated with hip replacements varied, with osteoarthritis associated with more than 80% of total hip replacements and fractures or congenital deformities being associated with more than 85% of partial hip replacements. There is some variation between diagnoses for the two databases. (Reference Table 4.11 PDF [29] CSV [30])
The 18-year mean age was about 66 years for total hip replacements and 77 for partial hip replacements, reflecting the different underlying diagnoses. Mean ages for both procedures show a slight decline over the time period, reflecting the younger age at which joint replacements are now considered. (Reference Table 4.12 PDF [31] CSV [32])
The mean length of stay for all hip replacements has shown a remarkable decline of at least 50% from 1992 through 2010. (Reference Table 4.13 PDF [33] CSV [34])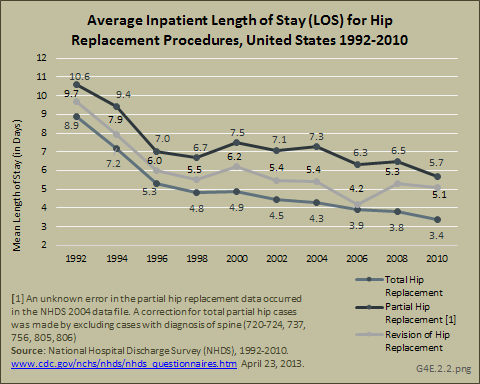
In spite of shorter hospital stays, mean hospital charges from 1998 through 2011 steadily increase for all hip replacements. Revision hip replacements are the most expensive, while total hip replacements are the least expensive. Total hospitalization charges for all hip replacements have tripled (in constant 2011 dollars) from $8.9 billion in 1998 to $27.8 billion in 2011, led by charges for total hip replacements. (Reference Table 4.14 PDF [35] CSV [36])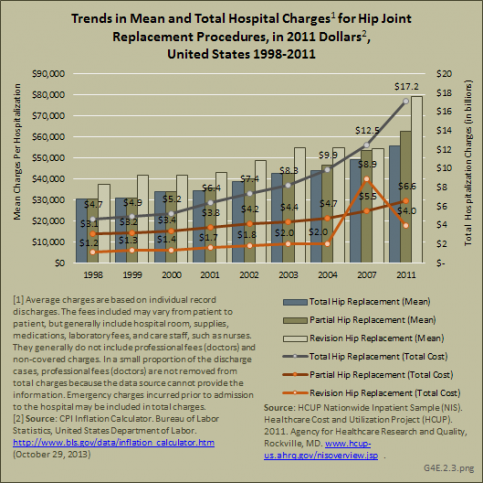
Most adults with total hip and revision hip replacements are routinely discharged home, but 20% to 30% are discharged to long-term care. Slightly higher rates of persons age 65 years and older go to long-term care. Among patients who undergo a partial hip replacement, nearly half (45%) go to long-term care, with three out of five who are 65 years and older with a partial hip replacement doing so. (Reference Table 4.15 PDF [37] CSV [38])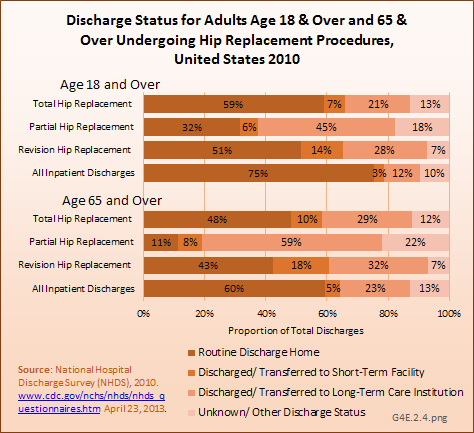
In 2010 and 2011, an estimated 67,000 to 76,000 shoulder replacement procedures were performed, comprising 5% to 6% of all joint replacement procedures. At the same time, an estimated 19,000 to 25,000 other joint replacement procedures were performed. This small group, which includes fingers, toes, wrist, ankle, and spine replacements, comprise less than 2% of all joint replacement procedures. As with hip and knee replacements, a majority of other joint replacement procedures occurred in females. (Reference Table 4.9 PDF [25] CSV [26])
Because many types of arthritis have a higher prevalence among older adults, expectations are that the current aging of the population will increase the prevalence and impact of AORC unless new means to lessen that impact are discovered in the near future. Projections of arthritis impact among adults are based on 2003 data adjusted for age distribution in the US population, but not for other potentially important factors such as the obesity epidemic and the possible increasing frequency of joint injuries.1 Doctor-diagnosed arthritis among adults is occurring as projected, with 52.5 million in 2010 to 2012 and 67 million expected by 2030. Arthritis-attributable activity limitations [39] among adults appear to be occurring at a faster pace than previously projected. This suggests the projected 25 million in 2030 will also be exceeded.1 Previous costs of arthritis have been driven by age-related increases in prevalence.2 Future costs of arthritis are likely to be driven higher by the same age-related increase in prevalence, but also from the increasing frequency of surgical interventions.
Juvenile arthritis (JA) is an umbrella term used to describe a number of autoimmune and inflammatory conditions that can develop in children.
The most common form of JA is Juvenile Idiopathic Arthritis (JIA), formally called juvenile rheumatoid arthritis (JRA) or juvenile chronic arthritis (JCA). Juvenile idiopathic arthritis is diagnosed in a child less than 16 years of age with at least 6 weeks of persistent arthritis. There are seven distinct subtypes, each having different symptoms and association to the child's autoimmunity and genetics.1 Certain subtypes are associated with an increased risk of inflammatory eye disease (uveitis). Understanding the differences in the various forms of JIA, their causes, and methods to better diagnose and treat these conditions in children is important for future treatment and prevention. Among all subtypes, 40% to 45% of children with JIA still have active disease after 10 years.2
Because of the various forms of JA, estimates of prevalence and incidence are difficult to identify. Overall estimates are that 300,000 children in the United States are diagnosed with JIA.3
In 2006, the CDC Arthritis Program finalized a case definition for ongoing surveillance of pediatric arthritis and other rheumatologic conditions (SPARC [40]) using the current ICD-9-CM diagnostically based data systems.
Analysis of recent national health care databases using the SPARC showed that 61,800 children age 17 years and younger were discharged from a hospital with any diagnosis of SPARC in 2011. Of those, 17,000 (more than one in four) had an admitting diagnosis of SPARC. Distribution was evenly split between males and females, but children age 6 years and younger were more likely to be hospitalized with a diagnosis of SPARC than older children, accounting for 45% with any SPARC diagnosis and half with an admitting SPARC diagnosis. Only a small number of children (3.2%) discharged with any diagnosis of SPARC had a diagnosis of juvenile idiopathic arthritis.1 Females accounted for 70% of discharges with a diagnosis of JIA, with 50% of the discharges for children age 13 to 17 years.
Hospital stays of nearly 9 days (mean of 8.6 days) were found for any diagnosis of SPARC. Children age 6 years and younger had a mean hospital stay nearly twice that of children ages 7 to 17 years, resulting in higher mean hospital charges. Children with a diagnosis of JIA had hospital stays of a mean of 3.6 days, with subsequently lower mean charges.
Total hospital charges associated with any diagnoses of SPARC in the population age 17 years and younger were $4.45 billion in 2012. (Reference Table 4.16 PDF [43] CSV [44])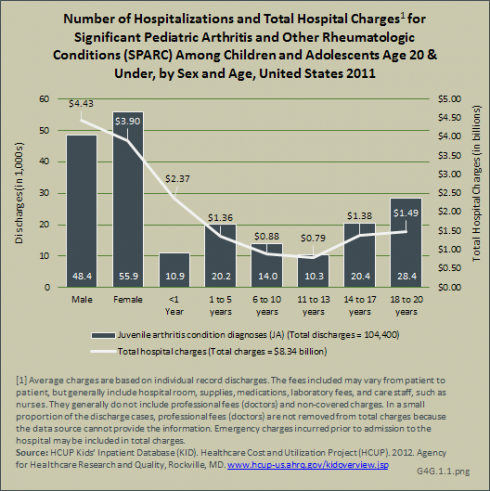
Emergency rooms saw 443,000 patients ages 1 day to 17 years with any diagnoses of SPARC in 2010. Among these patients, 5,400 had a primary diagnosis of JIA. Females and those ages 13 to 17 years were most likely to have juvenile idiopathic arthritis.
Due to smaller sample sizes in the currently available databases for physician office visits and outpatient clinics, outpatient visits for a diagnosis of SPARC in the juvenile population are difficult to quantify. Physician visits for treatment of JA for the years 2008 to 2010 averaged just over 1 million per year. Evenly split between males and females, 60% of the visits were for children ages 13 to 17 years. Because of small sample sizes, the number of visits with a diagnosis of JIA was unreliable.
Outpatient clinics saw 282,500 patients, on a yearly average, for these same years. Patterns for distribution by sex and age reflected that of other treatment sites. However, a larger share of these patients (17%) had a diagnosis of JIA.
From these data, an estimated 1.8 million outpatient visits for any diagnoses of SPARC occurred in those 1 day to 17 years of age in 2010. (Reference Table 4.16 PDF [43] CSV [44])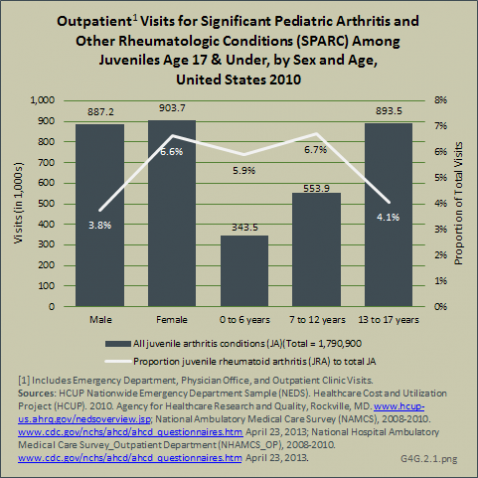
The Economic Cost [45] section of this report uses the Medical Expenditures Panel Survey (MEPS), a standard source of cost of illness estimates, to estimate the total direct and indirect costs of musculoskeletal conditions and selected categories of musculoskeletal conditions, as well as the incremental direct and indirect costs specifically attributable to the selected category. Total costs are all costs for a patient regardless of the condition responsible; incremental costs are those costs attributed to a specified condition.
There are several important points to remember here. First, for arthritis and other rheumatic conditions, MEPS requires the use of selected three-digit ICD-9-CM codes, using the three- and four-digit NADW AORC ICD-9-CM codes [1] to create a similar category called “arthritis and joint pain.” This approach provides the best available estimates of the costs of AORC. Additionally, costs estimates are per person and reported as mean per-person costs. To arrive at the estimated aggregate cost, the mean per-person cost is multiplied by the number of people affected, resulting in a total cost for conditions in the United States.
MEPS provides estimates of actual medical expenditures, meaning money changing hands, rather than medical charges, which are based on what is originally billed but rarely paid in full. Thus, the term direct costs as used here reflects actual medical expenditures.
For total medical expenditures, mean per-person expenditures for arthritis and joint pain increased from an average of $6,393 in the years 1996 to 1998 to an average of $9,556 from 2009 to 2011 in 2011 dollars. For incremental medical expenditures, mean per-person expenditures for arthritis and joint pain increased from an average of $654 in the years 1996 to 1998 to an average of $1,909 in 2009 to 2011 in 2011 dollars. The change in total mean expenditures was 50%, while incremental mean expenditures rose by 192%. (Reference Table 10.6 PDF [46] CSV [47])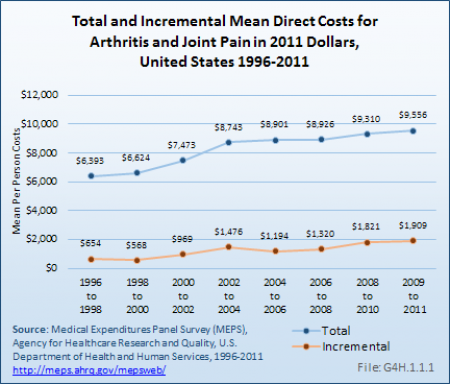
Mean per person direct costs include ambulatory care, inpatient care, prescriptions, and other health care costs. In 2011, ambulatory care and inpatient care each accounted for about a third of per person direct costs, with prescriptions accounting for another 25%. Over the past 15 years, prescription costs have seen the greatest change, rising nearly 60% per person in that time. Both inpatient and other health care costs went down by 20% and 23%, respectively. (Reference Table 10.4 PDF [48] CSV [49])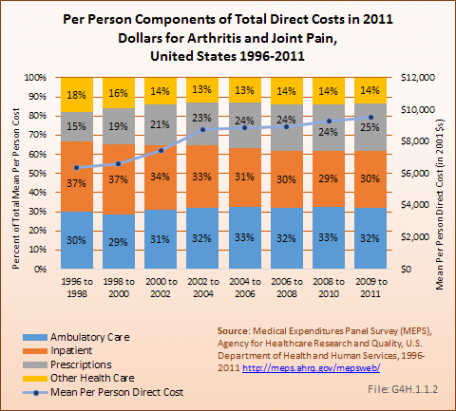
Total aggregate medical expenditures for people treated for all causes, including a diagnosis of arthritis and joint pain, in the United States increased from $185.2 billion in 1996 to 1998 to $580.9 billion in 2011 dollars, on average, for the years 2009 to 2011. Aggregate medical expenditures specifically attributed to arthritis and joint pain (incremental costs) in the United States increased from $18.9 billion in 1996 to 1998 to $116.1 billion in 2011 dollars, on average, for the years 2009 to 2011. While the increase over the 15-year period for total aggregate costs was more than 200%, the increase for incremental aggregate costs was greater than 500%. (Reference Table 10.6 PDF [46] CSV [47])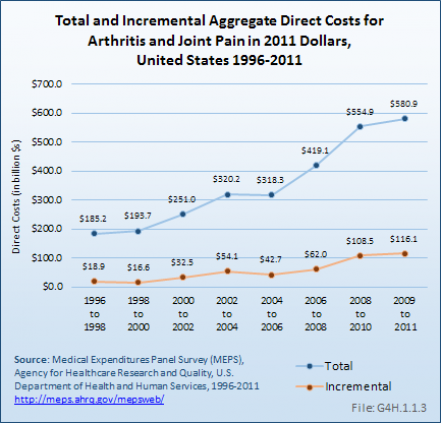
Mean and aggregate total and incremental direct and indirect costs for two types of arthritis using the annual average for years 2008 to 2011 MEPS data are calculated.
For total medical expenditures, mean per-person expenditures for osteoarthritis and allied disorders were $11,029 average for the years 2008 to 2011. Aggregate medical expenditures for the estimated 30.8 million persons with osteoarthritis and allied disorders in the United States averaged $340 billion in each of the years 2008 to 2011.
For incremental medical expenditures, mean per-person expenditures for osteoarthritis and allied disorders were $2,017 on average for the years 2008 to 2011. Aggregate incremental medical expenditures for the United States for osteoarthritis and allied disorders were $62.1 billion in each of the years 2008 to 2011. (Reference Table 10.13 PDF [50] CSV [51]).
For total medical expenditures, mean per-person expenditures for rheumatoid arthritis averaged $17,010 for the years 2008 to 2011. Aggregate medical expenditures for the estimated 1.04 million people with rheumatoid arthritis in the United States averaged $17.8 billion in each of the years 2008 to 2011.
For incremental medical expenditures, mean per person expenditures for rheumatoid arthritis averaged $6,428 for the years 2008 to 2011. Aggregate medical expenditures for the United States for rheumatoid averaged $6.7 billion in each of the years 2008 to 2011. (Reference Table 10.13 PDF [50] CSV [51])
Indirect costs as used in this report reflects estimates of earnings losses for people with a work history who are unable to work because of a medical condition. It does not reflect supplemental measures such as reduced productivity, worker replacement, or early retirement due to medical conditions.
Indirect costs are not estimated for the broad category of arthritis and joint pain.
For total earnings losses, mean per-person earnings losses attributed to osteoarthritis and allied disorders averaged $7,548 per year in 2008 to 2011. Aggregate earnings losses for the 16.1 million people in the workforce with osteoarthritis and allied disorders in the United States averaged $122 billion in each of the years 2008 to 2011.
For incremental medical expenditures, mean per-person earnings losses attributed to osteoarthritis and allied disorders averaged $4,951 per year in 2008 to 2011. Aggregate earnings losses for the United States due to osteoarthritis and allied disorders averaged $80 billion in each of the years 2008 to 2011.
Combining direct and indirect costs for osteoarthritis and allied disorders sums to total average costs of $461 billion, with incremental costs of $142 billion. (Reference Table 10.13 PDF [50] CSV [51])
For total earnings losses, mean per-person earnings losses attributed to rheumatoid arthritis averaged $13,886 per year in 2008 to 2011. Aggregate earnings losses for the estimated 900,000 persons in the workforce with rheumatoid arthritis in the United States averaged $12.3 billion per year in 2008 to 2011.
For incremental medical expenditures, mean per-person earnings losses attributed to rheumatoid arthritis averaged $8,684 per year in 2008 to 2011. Aggregate earnings losses for the United States due to rheumatoid arthritis averaged $7.7 billion in each of the years from 2008 to 2011.
Combining direct and indirect costs for rheumatoid arthritis sums to total costs of $30 billion, with incremental costs of $14.4 billion. (Reference Table 10.13 PDF [50] CSV [51])
Several data limitations exist for addressing AORC in the future. First, on October 1, 2015, ICD-10-CM will be required for use in clinical records; it is already in use for death records. The current National Arthritis Data Workgroup definition of AORC uses ICD-9-CM codes. Due to changes in conditions for the new codes, a direct translation cannot be made. This means a new definition of AORC or some similar concept will be needed for analyses using ICD-based data after that date. CDC will be working with ICD-10-CM translation experts and selected stakeholders to propose a draft standard ICD-10-CM–based definition, which will shared with the larger arthritis community to reach agreement on a new definition.
Second, there is a need for data on more specific conditions—for example, rheumatoid arthritis, SLE, psoriatic arthritis—to help drive clinical (eg, treatment, quality) and public health (eg, self-management education, safe physical activity) efforts. Electronic health records may prove helpful in creating valid measures.
Arthritis and other rheumatic conditions are not addressed with the same priority as many other chronic conditions, perhaps because such priorities are driven more by measures of mortality rather than measures for quality of life and disability. However, there is a growing policy interest in the role of multiple chronic conditions in health and health costs.1 Arthritis and other rheumatic conditions play a major role from this perspective, with about half of people with heart disease or diabetes and about a third of those with obesity also affected by arthritis and rheumatic conditions.2,3,4
There are widespread and consistent professional recommendations for most types of AORC that involve increasing self-management of the disease through education, physical activity, and achieving a healthy weight, but little progress is being made.1 Such behavior interventions offer evidence-based improvements to patients without the side effects seen with medications and other interventions. While most clinical settings are not set up to help patients achieve these recommendations effectively, increasing clinical/community linkages may offer a better approach. To see if provider referrals to community resources is a better solution, approaches such as the 1.2.3 Approach to Provider Outreach [53] and Spread the Word: Marketing Self-Management Education [54]Through Ambassador Outreach are being pilot tested in communities.
The Healthy People [55] project started with the 1979 Surgeon General’s Report, Healthy People [56]: The Surgeon General’s Report on Health Promotion and Disease Prevention. Healthy People 2020, the current program, has set nine arthritis objectives for the nation to achieve by 2020, but only limited progress has occurred with the current level of investments in interventions. Currently, four new objectives are being proposed in the Arthritis, Osteoporosis, and Chronic Back Conditions [57] topic area as part of a larger effort to ensure that chronic pain, as a condition separate from the original cause, is included in Healthy People 2020.
There is a need for more conveniently measured outcomes that are important to most people. Such outcomes include effects on work, activities, health-related quality of life, independence, and ability to keep doing valued life activities.
Research funding to develop and evaluate more effective clinical and public health interventions is relatively modest, given that arthritis is the most common cause of disability and is a large and growing problem, affecting 52.5 million adults now, and a projected 67 million by 2030.2 This is especially frustrating because even the evidence-based interventions we have now are not reaching the people who would benefit from them.
Although most adults with doctor-diagnosed arthritis are younger than 65 years and in the working years, the effect of their arthritis on employment and work, and the effect of reasonable workplace accommodations, have not been explored in depth. This is an urgent issue right now, and will continue to be an urgent issue as an aging workforce keeps working beyond age 65 years, as is anticipated.
Codes used in this analysis of AORC are based on the National Arthritis Data Workgroup ICD-9-CM diagnostic codes for arthritis and other rheumatic conditions.1 [[Link does not work -- please advise.]]
Osteoarthritis and Allied Disorders:
Osteoarthritis and allied disorders: 715
Rheumatoid Arthritis
Rheumatoid arthritis and other inflammatory polyarthropathies: 714
Gout and Other Crystal Arthropathies:
Gout: 274
Crystal arthropathies: 712
Joint Pain, Effusion and Other Unspecified Joint Disorders
Other unspecified arthropathies: 716.1, 716.3-716.6-716.9
Other and unspecified joint disorders: 719.0, 719.4-719.9
Spondylarthropathies:
AS/inflammatory spondylopathies: 720
Spondylosis and allied disorders: 721
Reiter’s disease: 99.3
Psoriatic arthopathy: 696.0
Fibromyalgia:
Myalgia and myositis unspecified: 729.1
Diffuse Connective Tissue Disease
Diffuse connective tissue disease: 710 [excl 710.0-.2]
Sicca syndrome (also called Sjögren's syndrome): 710.2
Systemic sclerosis (SSC, scleroderma): 710.1
Systemic lupus erythematosus (SLE): 710.0
Carpal Tunnel Syndrome:
Carpal tunnel syndrome: 354.0
Soft Tissue Disorders (excluding back):
Peripheral enthesopathies and allied disorders: 726
Other disorders of synovium/tendon/bursa: 727
Disorders of muscle/ligament/fascia: 728.0-728.3, 728.6-728.0
Rheumatism, unspecified and fibrositis: 729.0
Fascitis, unspecified: 729.4
Other Specified Rheumatic Conditions:
Syphilis of muscle: 95.6
Syphilis of synovium/tendon/bursa: 95.7
Gonococcal infection of joint: 98.5
Behcet’s syndrome: 136.1
Other disorders purine/pyrimidine metabolism: 277.2
Allergic purpura: 287.0
Cauda equina syndrome: 344.6
Brachial plexus/thoracic outlet lesions:353.0
Tarsal tunnel syndrome: 355.5
Polyneuropathy in collagen vascular disease: 357.1
Rheumatic fever w/o heart disease: 390
Rheumatic fever w/heart disease: 391
Cerebral arteritis: 437.4
Raynaud’s syndrome: 443.0
Polyarteritis nodosa and allied conditions: 446 [excl 446.5]
Arteritis, unspecified: 447.6
Arthritis associated with infections: 711
Arthropathy associated w/disorders classified elsewhere: 713
Specified arthropathies: 716.0, 716.2-716.8
Specified joint disorders: 719.2, 719.3
Polymyalgia rheumatica: 725
Links:
[1] https://bmus.latticegroup.com/2014-report/iik0/icd-9-cm-codes-arthritis-and-other-rheumatic-conditions
[2] http://www.cdc.gov/arthritis/data_statistics/case_definition.htm
[3] https://bmus.latticegroup.com/2013-report/definition/iv1
[4] https://bmus.latticegroup.com/docs/T4.1.pdf
[5] https://bmus.latticegroup.com/docs/T4.1.csv
[6] https://bmus.latticegroup.com/docs/T4.2.pdf
[7] https://bmus.latticegroup.com/docs/T4.2.csv
[8] https://bmus.latticegroup.com/docs/T4.3.pdf
[9] https://bmus.latticegroup.com/docs/T4.3.csv
[10] https://bmus.latticegroup.com/docs/T4.4.pdf
[11] https://bmus.latticegroup.com/docs/T4.4.csv
[12] https://bmus.latticegroup.com/docs/T4.5.pdf
[13] https://bmus.latticegroup.com/docs/T4.5.csv
[14] https://bmus.latticegroup.com/docs/T4.7.pdf
[15] https://bmus.latticegroup.com/docs/T4.7.csv
[16] http://www.cdc.gov/nchs/nhis.htm
[17] https://bmus.latticegroup.com/docs/T4.6.1.pdf
[18] https://bmus.latticegroup.com/docs/T4.6.1.csv
[19] https://bmus.latticegroup.com/docs/T4.6.2.pdf
[20] https://bmus.latticegroup.com/docs/T4.6.2.csv
[21] https://bmus.latticegroup.com/docs/T4.6.3.pdf
[22] https://bmus.latticegroup.com/docs/T4.6.3.csv
[23] https://bmus.latticegroup.com/docs/T4.8.pdf
[24] https://bmus.latticegroup.com/docs/T4.8.csv
[25] https://bmus.latticegroup.com/docs/T4.9.pdf
[26] https://bmus.latticegroup.com/docs/T4.9.csv
[27] https://bmus.latticegroup.com/docs/T4.10.pdf
[28] https://bmus.latticegroup.com/docs/T4.10.csv
[29] https://bmus.latticegroup.com/docs/T4.11.pdf
[30] https://bmus.latticegroup.com/docs/T4.11.csv
[31] https://bmus.latticegroup.com/docs/T4.12.pdf
[32] https://bmus.latticegroup.com/docs/T4.12.csv
[33] https://bmus.latticegroup.com/docs/T4.13.pdf
[34] https://bmus.latticegroup.com/docs/T4.13.csv
[35] https://bmus.latticegroup.com/docs/T4.14.pdf
[36] https://bmus.latticegroup.com/docs/T4.14.csv
[37] https://bmus.latticegroup.com/docs/T4.15.pdf
[38] https://bmus.latticegroup.com/docs/T4.15.csv
[39] https://bmus.latticegroup.com/2013-report/qwb-x1-quality-well-being-scale-nhis/iv3c
[40] http://www.cdc.gov/arthritis/data_statistics/case_definition/pediatric.htm
[41] http://www.arthritis.org/arthritis-facts/disease-center/juvenile-arthritis.php
[42] http://www.cdc.gov/arthritis/basics/childhood.htm
[43] https://bmus.latticegroup.com/docs/T4.16.pdf
[44] https://bmus.latticegroup.com/docs/T4.16.csv
[45] https://bmus.latticegroup.com/2014-report/x/economic-cost
[46] https://bmus.latticegroup.com/docs/T10007.6.pdf
[47] https://bmus.latticegroup.com/docs/T10007.6.csv
[48] https://bmus.latticegroup.com/docs/T10005.4.pdf
[49] https://bmus.latticegroup.com/docs/T10005.4.csv
[50] https://bmus.latticegroup.com/docs/T10014.13.pdf
[51] https://bmus.latticegroup.com/docs/T10014.13.csv
[52] http://www.hhs.gov/ash/initiatives/mcc/
[53] http://www.cdc.gov/arthritis/interventions/marketing-support/1-2-3-approach/index.html
[54] http://www.cdc.gov/arthritis/interventions/marketing-support/ambassador-outreach/index.html
[55] http://www.healthypeople.gov/
[56] http://profiles.nlm.nih.gov/NN/B/B/G/K/
[57] http://www.healthypeople.gov/2020/topics-objectives/topic/Arthritis-Osteoporosis-and-Chronic-Back-Conditions
[58] http://www.healthypeople.gov/2020/data-search/Search-the-Data?&f[0]=field_topic_area%3A3507
[59] http://www.cdc.gov/arthritis/data_statistics/arthritis_related_stats.htm
[60] http://www.cdc.gov/arthritis/data_statistics/pdf/arthritis_codes_2004.pdf
[61] http://www.cdc.gov/arthritis/data_statistics/pdf/idcm_codes_pediatric_arthritis.pdf